

8,000 schools use Gynzy
92,000 teachers use Gynzy
1,600,000 students use Gynzy
General
The world of chemistry is absolutely critical to everything we do, yet it is invisible to us. This lesson explores the "chemical language" of our world and teaches students to read and write chemical formulas, and understand why they are written the way they are.
Standards
NGSS: MS-PS1-1
Learning objective
Students will be able to understand the meaning of chemical formulas.
Introduction
Chemical formulas can look intimidating, but just like words, they are made up of letters (and numbers) that can be individually interpreted to create a larger meaning, or in this case, compound.
Instruction
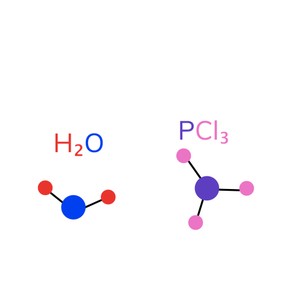
A chemical formula is a short way of writing the chemical symbols and numbers that represent substances, molecules, and compounds in chemistry. These abbreviations consist of a capital letter and often a lower case letter. Each chemical also has a corresponding atomic number and atomic mass that gives us more information about it. This information can be found in the periodic table of elements.
A noteworthy subset of chemicals is covalent molecules which are made up of nonmetal elements (groups 13-17 and hydrogen). Names of covalent compounds must include prefixes that indicate how many atoms of each element are present. Another subset is ionic compounds, which do not use prefixes in naming and which contain metals and nonmetals.
Quiz
Students will respond to ten true/false, multiple-choice, and short answer questions.
Closing
Students are shown a molecule and given the names of the makeup of it, and asked to write the correct chemical formula. Is the result covalent or ionic? What is the full name of the molecule?
The online teaching platform for interactive whiteboards and displays in schools
Save time building lessons
Manage the classroom more efficiently
Increase student engagement
Discover more!
About Gynzy
Gynzy is an online teaching platform for interactive whiteboards and displays in schools.
With a focus on elementary education, Gynzy’s Whiteboard, digital tools, and activities make it easy for teachers to save time building lessons, increase student engagement, and make classroom management more efficient.